There is still debate about the effectiveness of Continuous Subcutaneous Insulin Infusion (CSII). Several studies indicate that improvements in glycaemic control or quality of life strongly depend on psychological, educational and motivational factors. We analysed decisions/reasons for discontinuation of pump use in children and youth with type 1 diabetes.
In a retrospective cross-sectional study in a single center (Diabeter, currently 62% CSII-use), clinical data between 2007 and 2018 were extracted from EHRs of people with type 1 diabetes aged 0-25 years. Reasons for discontinuation (practical, emotional and clinical/technical) were assessed by questionnaire (19% response rate).
Key findings:
- People with type 1 diabetes reported dissatisfaction/disappointment with/by CSII (device influencing body image, 66%; increased confrontation with type 1 diabetes, 58%)
- Discontinuation due to clinical factors were mostly advised by diabetes team members due to low daily insulin dose (skipping insulin boluses), minimal glucose measurements (leading to worse glycaemic control), and skin infections/irritations (58%)
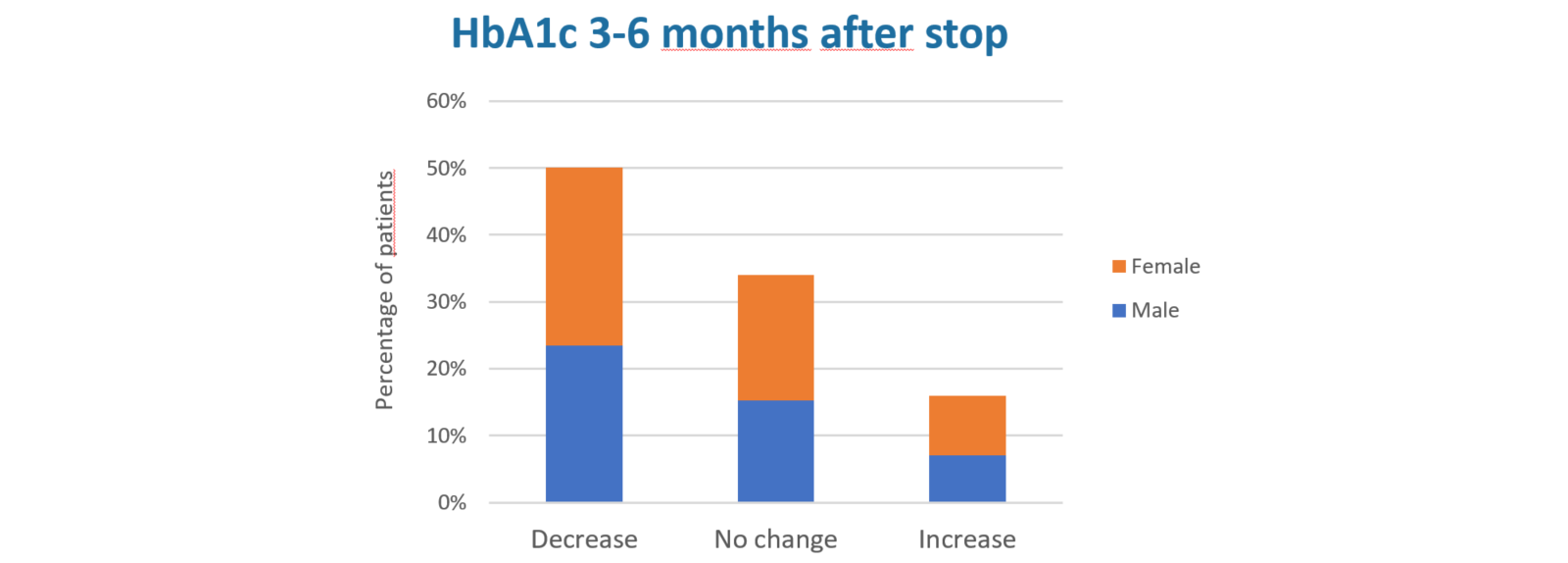
- Of people with type 1 diabetes with available HbA1c data before (HbA1c 9.8%) and 3-6 months post-discontinuation, 54% showed improved HbA1c (-1.7%) post-discontinuation: 34%/12% showed no change/increased HbA1c (+1.1%: here factors other than insulin delivery likely play a determining role)
- The researchers concluded that “our data identify a subgroup with worsening glycaemic control reporting negative experiences” and that “using an approach including a realistic evaluation of patient expectations and preferences” they hope to identify people at risk for discontinuation of CSII and to provide them with proper support (training and education) before and during CSII